if you were going to graphically determine the activation energy of this reaction what points
Activation Energy
Before going on to the Activation Free energy, permit's wait some more at Integrated Rate Laws. Specifically, the use of first order reactions to calculate Half Lives.
Permit's review before going on...
Integrated forms of rate laws:
In order to empathise how the concentrations of the species in a chemical reaction change with fourth dimension it is necessary to integrate the rate constabulary (which is given as the time-derivative of i of the concentrations) to find out how the concentrations change over fourth dimension.
1. Commencement Order Reactions
Suppose nosotros accept a beginning gild reaction of the form, B + . . . . → products. Nosotros tin can write the rate expression as rate = -d[B]/dt and the charge per unit police every bit rate = thousand[B]b . Set the two equal to each other and integrate information technology every bit follows:
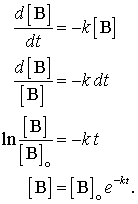
The outset order charge per unit constabulary is a very important rate law, radioactive disuse and many chemical reactions follow this charge per unit law and some of the language of kinetics comes from this law. The final Equation in the serial higher up iis called an "exponential disuse." This class appears in many places in nature. One of its consequences is that information technology gives ascension to a concept called "half-life."
Half-life
The half-life, usually symbolized past t1/2, is the time required for [B] to drop from its initial value [B]0 to [B]0/2. For Example, if the initial concentration of a reactant A is 0.100 mole L-ane, the one-half-life is the time at which [A] = 0.0500 mole L-1. In general, using the integrated form of the first order rate constabulary we discover that:
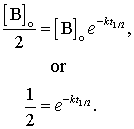
Taking the logarithm of both sides gives:
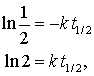
The half-life of a reaction depends on the reaction lodge.
Offset order reaction: For a offset club reaction the half-life depends only on the rate constant: 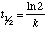
Thus, the half-life of a first order reaction remains abiding throughout the reaction, even though the concentration of the reactant is decreasing.
Second order reaction: For a second club reaction (of the grade: rate=one thousand[A]two) the half-life depends on the changed of the initial concentration of reactant A: 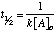
Since the concentration of A is decreasing throughout the reaction, the half-life increases as the reaction progresses. That is, it takes less time for the concentration to driblet from 1M to 0.5M than it does for the drib from 0.5 M to 0.25 K.
Here is a graph of the ii versions of the half life that shows how they differ (from http://world wide web.brynmawr.edu/Acads/Chem/Chem104lc/halflife.html)
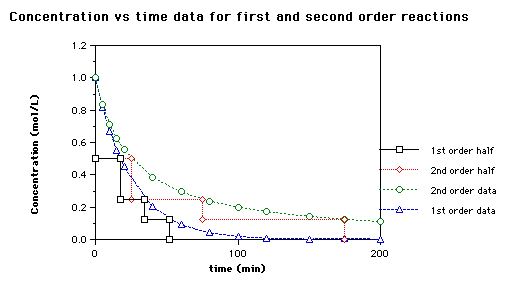
Allow'south try a simple problem: A beginning order reaction has a rate constant of 1.00 s-i. What is the half life of the reaction?
Since the reaction is first guild we need to utilise the equation: t1/2 = ln2/thou
tane/2 = ln2/(1.00 s-1) = 0.6931 s
Now let'south try a harder problem:
The half-life of North2O5 in the first-order decomposition @ 25°C is 4.03×x4s. What is the rate constant? What percentage of Northward2O5 will remain later one twenty-four hour period?
![]()
At that place are 24 hours * 60 min/60 minutes * threescore sec/min = 8.64×10four due south in a solar day
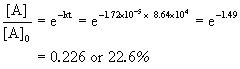
So 22.half-dozen % remains after the end of a mean solar day.
At present, Activation Free energy:
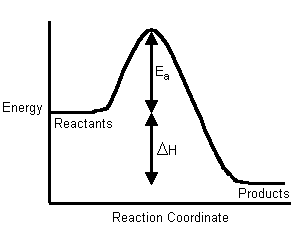
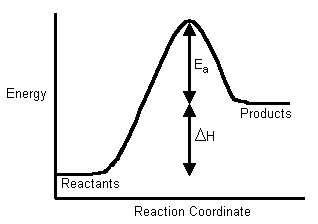
The Activation Energy (Eastwarda) - is the energy level that the reactant molecules must overcome before a reaction tin occur.
You probably retrieve from CHM1045 endothermic and exothermic reactions:
 |  |
|---|
In order to calculate the activation energy nosotros need an equation that relates the rate abiding of a reaction with the temperature (free energy) of the organization. This equation is called the Arrhenius Equation:
![]()
Where Z (or A in modern times) is a abiding related to the geometry needed, m is the rate constant, R is the gas constant (8.314 J/mol-K), T is the temperature in Kelvin. If we rearrange and have the natural log of this equation, we can then put it into a "directly-line" format:
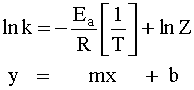
So now we can use it to calculate the Activation Energy past graphing lnk versus one/T.
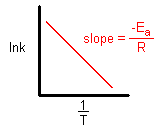
When the lnk (charge per unit constant) is plotted versus the changed of the temperature (kelvin), the slope is a direct line. The value of the gradient (yard) is equal to -Ea/R where R is a constant equal to viii.314 J/mol-Thousand.
"Two-Signal Form" of the Arrhenius Equation
The activation free energy can besides be found algebraically by substituting ii rate constants (k1, k2) and the two respective reaction temperatures (T1, T2) into the Arrhenius Equation (2).



Substracting equation (4) from equation (3) results in
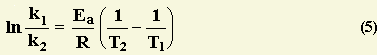
Rerrangement of equation (5) and solving for E a yields

Permit'due south try a problem:
The rate constant for the reaction Htwo(g) + I2(g) ---> 2HI(g) is 5.4 ten x-4Grand-1s-one at 326oC.
At 410oC the rate abiding was found to be 2.8 x 10-2 Grand-1s-1.
Calculate the a) activation energy and b) high temperature limiting rate constant for this reaction.
Answer:
All reactions are activated processes. Rate constant is exponentially dependent on the Temperature
We know the rate constant for the reaction at two different temperatures and thus nosotros tin can calculate the activation energy from the above relation. Get-go, and always, catechumen all temperatures to Kelvin, an absolute temperature scale. And then simply solve for Ea in units of R.
ln(5.4 x 10-4M-1s -1/ two.eight x 10-ii M-1s-i) = (-Ea /R ){1/599 K - 1/683 K}
-3.9484 = - Due easta/R {2.053 x 10-4 K-i}
Ea= (1.923 x 104 K) (viii.314 J/M mol)
Due easta= one.60 x 10v J/mol
Now that we know Ea, the pre-exponential factor, A, (which is the largest rate constant that the reaction can possibly have) tin exist evaluated from any mensurate of the absolute rate constant of the reaction.
and so
five.4 x ten-4M -1s-1 =
A exp{-(1.60 x ten5 J/mol)/((8.314 J/Chiliad mol)(599K))}
(five.4 10 x-4K-anedue south-1) / (ane.141x10-xiv) = iv.73 x xtenM-ones-1
The infinite temperature rate constant is iv.73 x x10Thousand-onedue south-one
Endeavour i with graphing:
Variation of the rate constant with temperature for the first-lodge reaction 2N2O5(g) -> 2NiiO4(m) + Otwo(k) is given in the following table. Determine graphically the activation energy for the reaction.
| T (1000) | k (south-1) |
|---|---|
| 298 | 1.74 x 10-5 |
| 308 | six.61 x 10-five |
| 318 | 2.51 x x-4 |
| 328 | vii.59 10 x-4 |
| 338 | two.40 x x-3 |
Respond:
Graph the Data in lnk vs. 1/T. It should issue in a linear graph.
The activation energy can be calculated from slope = -Ea/R. The value of the slope is -8e-05 and then:
-8e-05 = -Ea/8.314 --> Ea = 6.65e-4 J/mol
Source: https://www.chem.fsu.edu/chemlab/chm1046course/activation.html
Post a Comment for "if you were going to graphically determine the activation energy of this reaction what points"